
A standard sake may contain 10-29 ppm sodium and 42-185 ppm potassium. The method can normally be used for the determination of sodium, magnesium, potassium and calcium in precipitation within the range 0.01-2 mg/l, but this will depend to a certain degree on the commercial instruments used. A standard table wine may contain 5-50 ppm sodium and 400-1200 ppm potassium. 4.6.1 Determination by flame atomic spectroscopy (AAS or AES) 4.6.1.1 Field of application. We find that errors in these rates cannot remove the instability, but they do dramatically affect the range in parameter space over which it forms. Sodium and potassium are important components of wines and are routinely determined to check the integrity and quality of a wine. We investigate the effects of this estimated error for the well-known thermal instability of photoionized gas. Potassium K 1.1 20 40 Sodium Na 0.12 2 5 Strontium Sr 0.0019 0.03 0.07 Thallium Tl 0.16 2 6 Vanadium V 0.014. (LLNL), Livermore, CA (United States) Sponsoring Org.: USDOE National Nuclear Security Administration (NNSA) OSTI Identifier: 1823223 Report Number(s): LLNL-JRNL-761173 Journal ID: ISSN 1538-4357 950243 TRN: US2215610 Grant/Contract Number: AC52-07NA27344 Resource Type: Journal Article: Accepted Manuscript Journal Name: The Astrophysical Journal (Online) Additional Journal Information: Journal Volume: 881 Journal Issue: 2 Journal ID: ISSN 1538-4357 Publisher: IOP Publishing Country of Publication: United States Language: English Subject: 79 ASTRONOMY AND = n=0 DR rates. Publication Date: Research Org.: Lawrence Livermore National Lab. Clarendon Lab., Oxford (United Kingdom).Imperial College, London (United Kingdom).Atomic Weapons Establishment (AWE), Reading (United Kingdom) The time-resolved emission spectrum of the neutral atomic potassium (K i) was recorded in the 7007000 cm-1 region using the Fourier transform infrared.Although intended to be sodium-free, salt substitutes contain small amounts of NaCl as an impurity. fumaric acid, calcium hydrogen phosphate, or potassium tartrate also may be present. 
of Michigan, Ann Arbor, MI (United States) NIST Physical Measurement Laboratory, Handbook of Basic Atomic Spectroscopic Data. Atomic emission occurs when a valence electron in a higher energy atomic orbital returns to a lower energy atomic orbital. (LLNL), Livermore, CA (United States) Univ. E = K α – K α for Si (where Si line energy = 1.Sum peaks (two X-ray photons arriving at the detector at the same time)Įscape peaks (Si in the detector absorbing some of the energy from a X-ray) Compton peaks from X-ray source target (typically at energies Rayleigh peaks from X-ray source target (typically Ag L α, L β).A light beam is directed through the flame to a monochromator, and then to a detector that measures the amount of light absorbed by the sample at a specific wavelength, determined by the element of interest. Bremsstrahlung (more prominent in less dense samples) Atomic potassium was generated by pulsed irradiation of potassium halide. 2.1 In flame atomic absorption spectrometry, water is atomized by aspiration into a flame.Journal of Physics B: Atomic, Molecular and Optical Physics. atomic emission spectra and the electronic structure of atoms. Peak overlaps arising from the presence of multiple elements in the sample and limited detector resolution Sub-doppler modulation spectroscopy of potassium for laser stabilization. Potassium, and less reactive Interisting Facts Its the sixth most used metal. 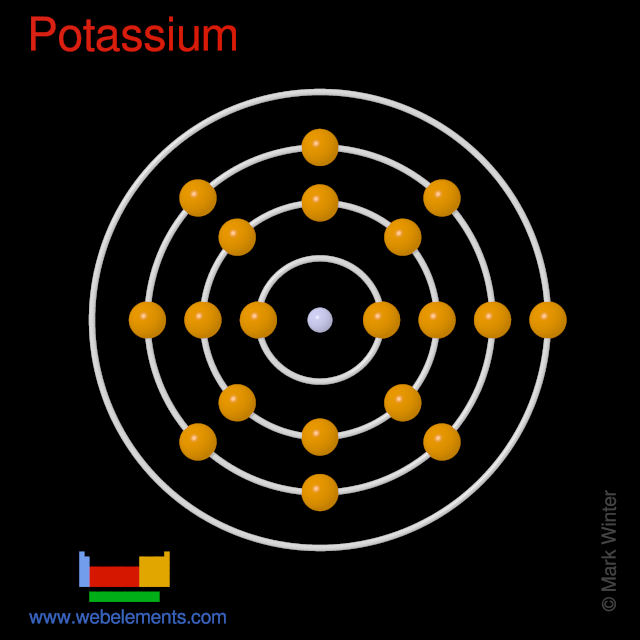
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
